Community Acquired Pneumonia
Community Acquired Pneumonia (CAP)
Common causative organisms according to site of care
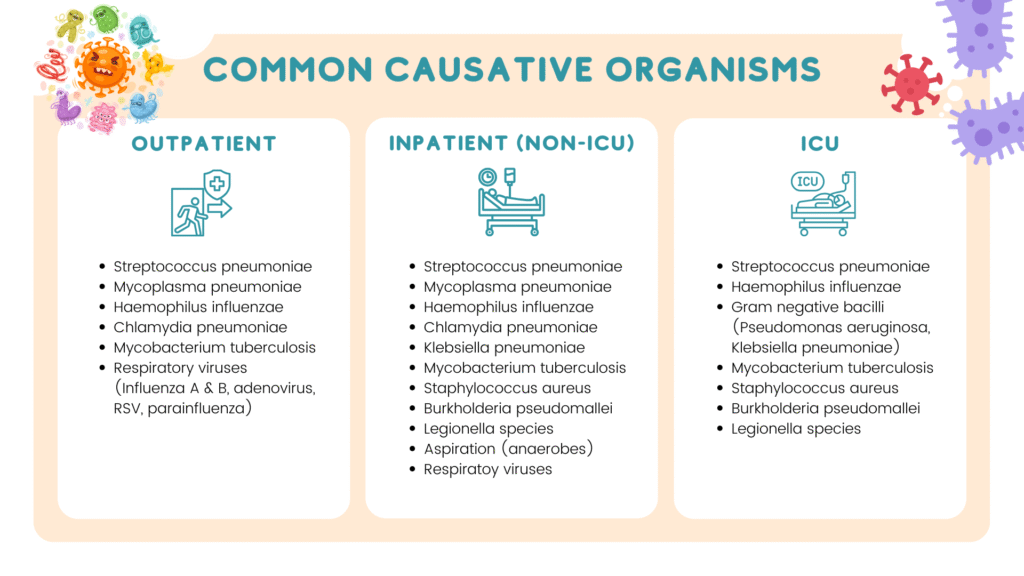
Burkholderia pseudomallei should be considered especially if the patient has diabetes mellitus.
Gram negative bacilli (Enterobacteriaceae & Pseudomonas may be the cause in those who have had antimicrobial treatment or who have pulmonary comorbitites.
Regarding pseudomonal infection
Strong risk factors
- Prior respiratory infection or isolation of Pseudomonas aeruginosa
- Hospitalization with receipt of IV antibiotics in past 3 months
- Detection of gram-stain rods on a good quality sputum gram stain
Other risk factors
- Recent hospitalization or stay in a long-term care facility.
- Recent antibiotic use
- Frequent COPD exacerbations requiring glucocorticoid/antibiotic
- Structural lung diseases (e.g. bronchiectasis)
- Immunosuppressed.
Deciding site of treatment
When treating CAP especially in primary care setting, it is important to decide on the site of treatment that is to treat as outpatient or patient requires referral to secondary/tertiary centre (general ward/ICU)
The decision can often be made by:
1. Risk stratification according to
- Presence of co-morbid conditions.
- Severity (taking into account physical findings, CXR, & lab findings)
- Presence of identified clinical risk factors for drug-resistant & unusual pathogen
2. The local epidemiology & resistance pattern.
Prognostic scoring tools
In addition to the factors mentioned above, there are few prognostic scoring tools that may be used to support our decision on site of treatment.
These include:
- Pneumonia severity index (PSI) – useful for identifying low-risk patient that can be safely treated as outpatient.
- CURB-65/CRB-65
- SMART-COP
The American Thoracic Society (ATS) guideline recommends PSI over CURB-65 as PSI is able to identify larger proportions of low-risk patients that can be treated as outpatient. In regards to CURB-65, there is paucity of evidence regarding its effectiveness/safety as decision aid in guiding initial site of treatment. Nevertheless, NICE guideline do recommend using CURB-65/CRB-65 for pneumonia severity assessment.
In our Malaysia NAG guidelines, CURB-65/CRB-65 and SMART COP are recommended scoring tools to aid in decision making on initial site of treatment.
However, in the ATS guideline, SMART COP is only used when patient has already been admitted to the hospital and it is used to identify patients who need vasopressor &/or mechanical ventilation and thus ICU admission.
Among these scoring tools, CRB-65 might be more suitable for primary care setting as it does not require any lab results/imaging.
It is important to keep in mind that these are used to support but not replace clinical decision making.

PSI and SMART COP are not listed in detail here as they require lab tests that may not be available in primary care setting.
ATS criteria for severe pneumonia
Severe CAP is defined as the presence of one major criteria or 2 minor criteria.
Major criteria
- Septic shock with need of vasopressors
- Respiratory failure requiring mechanical ventilation
Minor criteria
- Respiratory rate ≥ 30 breaths/min
- PaO2/FiO2 ratio ≤ 250
- Multilobar infiltrates on CXR
- Confusion/disorientation
- Uremia (BUN ≥ 20 mg/dl)
- Leukopenia (White blood cell < 4,000 cells/μL) – due to infection alone and not other cause (e.g. chemotherapy)
- Thrombocytopenia (Platelet < 100,000/μL)
- Hypothermia (Core temperature < 36 °C)
- Hypotension requiring aggressive fluid resuscitation.
Regarding blood C&S, sputum gram stain and C&S
Recommended only to take if :
- Severe CAP
- Being empirically treated for MRSA or P.aeruginosa
- Previously infected with MRSA or P. aeruginosa, especially those with prior respiratory tract infection
- Were hospitalized & received parenteral antibiotics, whether during the hospitalization event or not, in the last 90 days.
Most are strong recommendation but with low quality of evidence.
Whether to culture patients or not should be determined by individual clinicians based on clinical presentation, local etiological considerations, & local antimicrobial stewardship process.
Treatment 💊
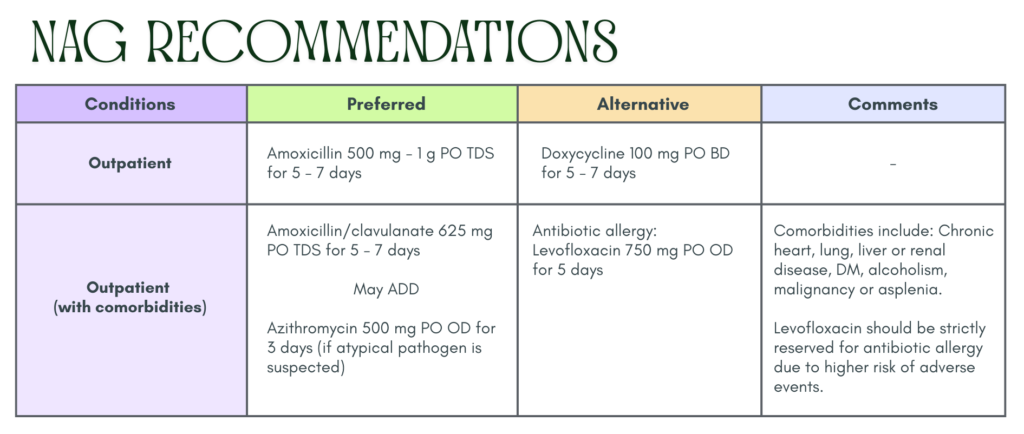
Inpatient management are not discussed here. Kindly refer to our Malaysia NAG guidelines if you would like to know more.
ATS treatment guideline
Similar to our Malaysia NAG guidelines
Outpatient (without comorbidities)
- Amoxicillin 1 g TDS or
- Doxycycline 100 mg BD or
- Macrolide (Azithromycin 500 mg D1 then 250 mg OD or clarithromycin 500 mg BD or clarithromycin extended release 1000 mg OD) monotherapy only in areas with pneumococcal resistence to macrolides < 25%
Outpatient (with comorbidities)
Augmentin 625 mg TDS or 875/125 mg BD or 2000 mg/125 mg BD or cephalosporin (cefpodoxime 200 mg BD or cefuroxime 500 mg BD)
+
Macrolide (Azithromycin 500 mg D1 then 250 mg OD or clarithromycin 500 mg BD or clarithromycin extended release
1000 mg OD)
OR
Monotherapy with respiratory fluoroquinolone (levofloxacin 750 mg OD. Moxifloxacin 400 mg OD or gemifloxacin 320 mg OD)
Note on corticosteroids
Routine corticosteroids administration are not recommended in the treatment of CAP. May be considered in refractory septic shock (in accordance to the Surviving Sepsis Campaign)
Duration of treatment ⌚
Antibiotic therapy should be continued until the patient achieve stability & for no less than a total of 5 days.
Failure to achieve clinical stability within 5 days is associated with higher mortality & worse clinical outcomes.
Longer course of antibiotic therapy are recommended for:
- Pneumonia complicated by meningitis, endocarditis, & other deep-seated infection.
- Infection with other, less common pathogens.
Routine follow up CXR is not suggested if symptoms have resolved within 5 to 7 days.
In adults with CAP who test positive for influenza
Antiinfluenza treatment is recommended to be started independent of duration of illness before diagnosis regardless of inpatient or outpatient (though quality of evidence and recommendation is stronger for inpatient).
Standard antibacterial treatment is recommended to be prescribed in those with clinical & radiographic evidence of CAP who test positive for influenza (especially in severe CAP)
If there is no evidence of a bacterial pathogen (including a low procalcitonin level) & early clinical stability, consideration could be given to earlier discontinuation of antibiotic treatment at 48 – 72 hours.
References
- Metlay, J. P., Waterer, G. W., Long, A. C., Anzueto, A., Brozek, J., Crothers, K., Cooley, L. A., Dean, N. C., Fine, M. J., Flanders, S. A., Griffin, M. R., Metersky, M. L., Musher, D. M., Restrepo, M. I., & Whitney, C. G. (2019). Diagnosis and Treatment of Adults with Community-acquired Pneumonia. An Official Clinical Practice Guideline of the American Thoracic Society and Infectious Diseases Society of America. American journal of respiratory and critical care medicine, 200(7), e45–e67. https://doi.org/10.1164/rccm.201908-1581ST
- Ministry of Health Malaysia. (2024). National antimicrobial guideline (NAG): Section A11 – Respiratory infections. Pharmaceutical Services Programme, Ministry of Health Malaysia.
