Androgenetic alopecia

Androgenetic Alopecia (AGA)
The Normal Hair Cycle
To understand about the pathophysiology of androgenetic alopecia, it is best to learn first about the normal hair cycle.
Our normal hair cycle generally consists of 4 phases: i. Anagen phase; ii. Catagen phase; iii. Telogen phase and iv. Exogen phase as illustrated below.
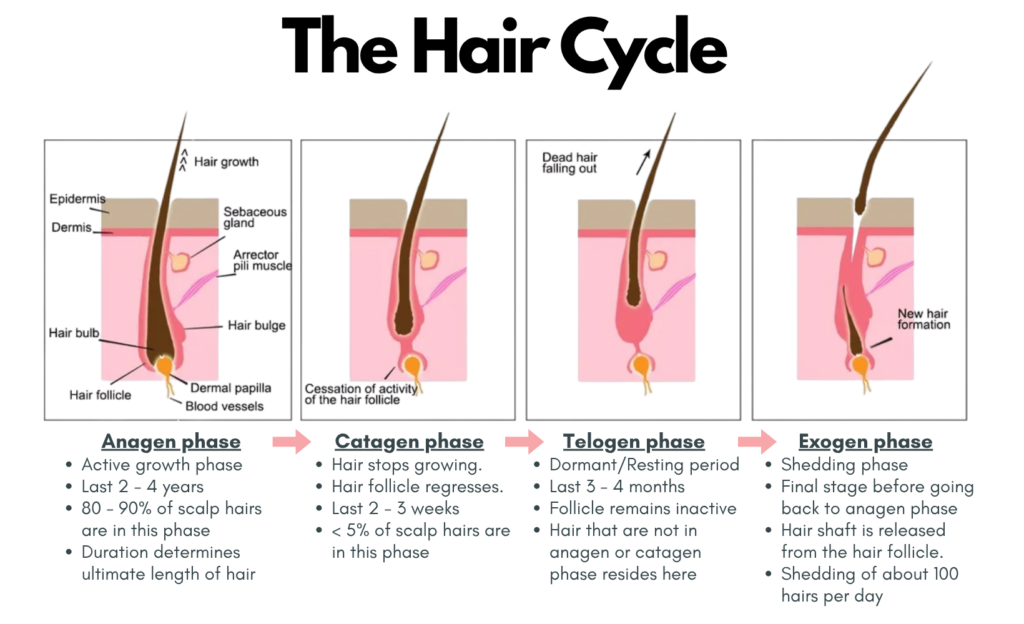
Pathophysiology of Androgenetic Alopecia
AGA is a genetically predetermined disorder characterized by an excessive response to androgens.
The condition is polygenic, involving multiple genes inherited from both parents. A paternal history of balding is associated with 5-6 fold increased risk.
In a person with AGA,
- The affected scalp areas show ⬆ activity of the enzyme 5⍺-reductase inhibitor & higher density of androgen receptors.
- Testosterone is converted into the more potent dihydrotestosterone (DHT) via 5⍺-reductase inhibitor (primarily the type 2 isoform) which subsequently activates the androgen receptors.
- This leads to :
a) Follicular miniaturization – progressive shrinkage of hair follicles –> shorter & thinner which may fail to penetrate the epidermal layer. Eventually, the thick terminal hairs are gradually replaced by fine vellus hairs.
b) Hair cycle alteration – shorter anagen phase & longer telogen phase –> ⬇ anogen : telogen ratio from ~12 : 1 (normal) to as low as ~5 : 1.
Patterns of Hair Loss
Male Pattern Hair Loss (MPHL)
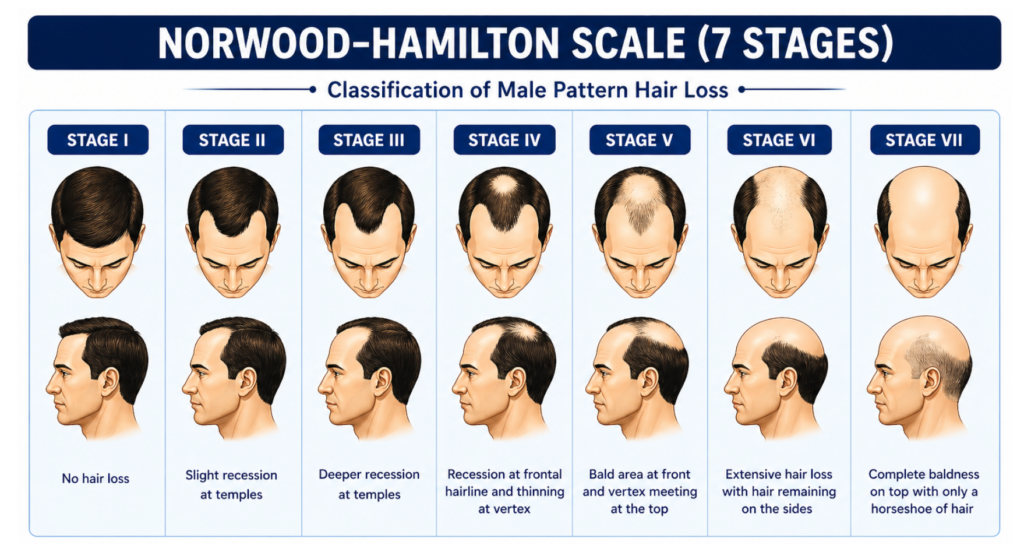
In male, the hair loss typically begins with bitemporal recession of the frontal hairline, followed by involvement of the vertex (the crown of the scalp). Hair loss is most prominent in these specific regions.
While some men may progress rapidly to complete baldness, others maintain a patterned thinning.
Stages of male pattern hair loss can be classified using the Norwood-Hamilton Scale which consists of 7 stages as illustrated above.
Female Pattern Hair Loss (FPHL)
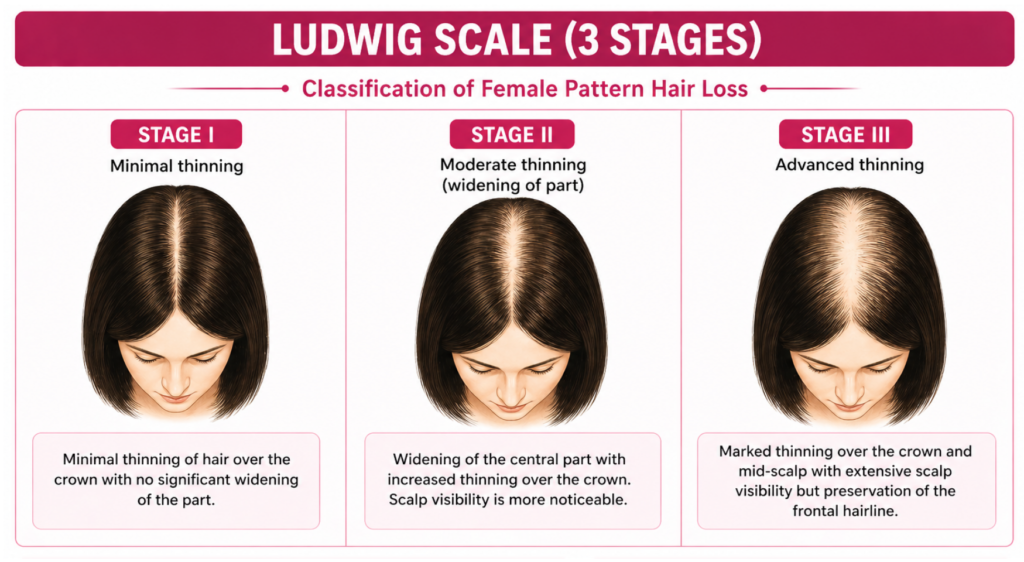
In female, the frontal hairline is typically spared. Instead, there is diffuse thinning concentrated on the head.
This loss is often first noticed as a wider appearance of the center part.
Women rarely experience complete baldness.
Female pattern hair loss can be classified using the Ludwig Scale which consists of 3 stages as shown above.
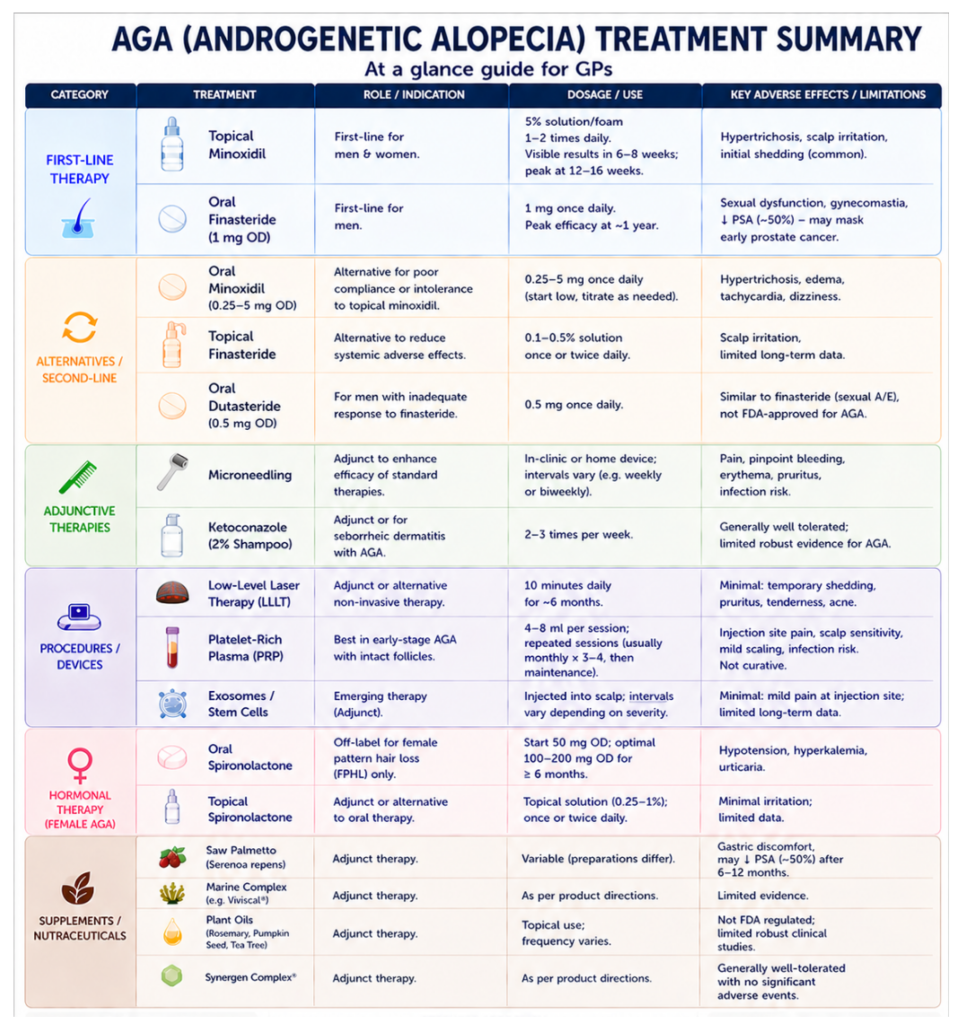
Treatment
There are 3 treatment options that are FDA-approved for AGA, these are: Topical Minoxidil, Oral Finasteride and Low-Level Light Therapy.
Nevertheless, there are adjuvant or off label treatment that has been used for the treatment of AGA.
We will be discussing briefly on the different treatment modalities for AGA.
AGA treatment modalities
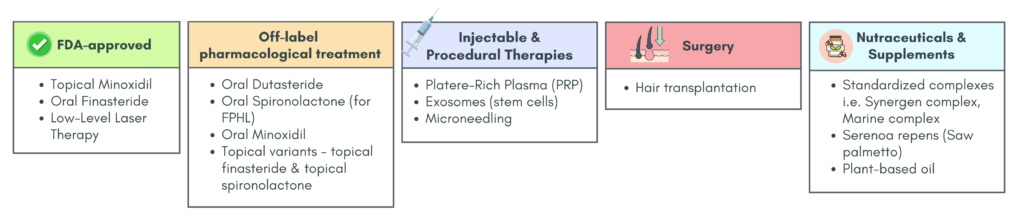
a) FDA-approved
Topical Minoxidil
Minoxidil is a prodrug that requires conversion to its active form, minoxidil sulfate, by sulfotransferase enzymes (SILT1A) within the hair follicle.
Clinical response varies depending on follicular sulfotransferase enzyme activity.
Onset of effect: ~6–8 weeks
Maximal response: ~12–16 weeks (may plateaus thereafter)
MOA:
- Vasodilation: Opens ATP-sensitive K⁺ channels → ↑ scalp blood flow and nutrient delivery
- Anti-inflammatory: ↓ pro-inflammatory cytokines (e.g. IL-2)
- Hair cycle modulation: Promotes anagen phase entry and prolongation
- Growth signaling: ↑ Wnt/β-catenin pathway and VEGF expression
Formulations:
- For men: 2% & 5% solution, 5% foam
- For women: 2% solution, 5% foam
Application:
- Apply once to twice daily (commonly BD) to affected areas.
- Consistent, long-term use is required to maintain effect.
Caution:
Transient increase in hair shedding may occur early in the treatment due to alterations of the hair cycle such that the hair follicle enters the anagen phase prematurely, thus passing through a shorter telogen phase. Typically self-limiting.
Adverse effects:
- Most common – Hypertrichosis
- Others: headache, irritant contact dermatitis (pruritus & scaling)
- Overall well tolerated with good safety profile.
Oral Finasteride
MOA: Selective 5a-reductase type II inhibitor –> reduce conversion of testosterone to DHT.
Dose: 1mg/day
Efficacy:
- Gradual response; peak effect ~ 1 year
- Continued improvement may occur beyond 1 year.
- Requires long-term maintenance to sustain hair regrowth.
Adverse effects:
- Sexual dysfunction (main concern as it affects compliance) – ⬇ libido, erectile dysfunction, ejaculatory dysfunction
- Post finasteride syndrome – sexual side effects + neuropsychiatric effects (i.e. depression)
– Often reversible, but pay persist in a subset of patients. - Others – gynecomastia, muscle atrophy, orthostatic hypotension
Caution (prostate cancer consideration)
While it reduces overall prostate cancer risk, it may be associated with an increased detection of high-grade disease, possibly due to diagnostic bias rather than true increased risk.
The apparent ↑ detection of high-grade cancer may be due to:
- ↓ prostate volume leading to improved biopsy accuracy
- ↑ PSA sensitivity
Nevertheless, patients still be informed of these historical controversies and continue to be regularly monitored for prostate health while using the medication
Monitoring includes:
- Perform baseline prostate assessment (e.g. DRE)
- Check PSA at baseline after ≥6 months, then monitor periodically
- Interpret PSA with caution (values may be reduced)
Contraindication: women of childbearing age (teratogenic – causing ambiguous genitalia in male fetus)
Low Level Laser Therapy
Non-invasive light-based therapy for AGA.
Can be used at home or in clinic
MOA:
- Uses light with wavelengths between 600 and 1100 nm to stimulate hair regrowth by promoting anagen-phase reentry of telogen hair follicles, prolonging duration of anagen phase & prevention of premature catagen transition.
- Red light absorption by cytochrome c oxidase (CCO) in mitochondria –> photodissociation of inhibitory nitric oxide (NO) –> increased ATP production, reactive oxygen species modulation, & transcription factor induction.
Clinical use:
- Adjunct to minoxidil/finasteride.
- Alternative to patients intolerant to medications.
Approved LLLT device for AGA: Lasermax Haircomb, The Capillus laser cap
Treatment: ~ 10 minutes daily for 6 months.
Adverse effects were minimal. May include temporary hair shedding, pruritus, tenderness & acne.
b) Off-label pharmacological treatment
1. Oral Dutasteride
MOA: Inhibits both type I and type II 5⍺-reductase.
Generally more potent (in blocking DHT & promoting hair growth) than finasteride, but is used off-label
Dose: available as 2.5 mg and 5 mg doses once daily – both of which have shown superior efficacy to finasteride 5 mg.
May be an effective therapeutic option in patients who fail oral finasteride.
Adverse effects (similar to finasteride): Sexual dysfunction – decreased libido, ejaculatory dysfunction, gynecomastia
2. Oral Spironolactone – used off-label for Female Pattern Hair Loss (FPHL) only.
An aldosterone antagonist (anti-androgen properties)
MOA:
- ↓ testosterone production in the adrenal gland by affecting the 17a-hydroxylase and desmolase.
- Also competitively inhibits the androgen receptor.
Dosage: Start 50 mg OD –> Titrate to 100 – 200 mg OD for at least 6 months to see clinical improvement.
Adverse effects: hypotension, hyperkalemia, occasional urticaria.
3. Oral Minoxidil
MOA: potent vasodilator (originally for resistant hypertension.)
Dose: 0.25 mg – 5 mg OD (Lower than antihypertensive dose of 10–40 mg/day)
Alternative option for those with poor adherence or intolerant to adverse effect of topical minoxidil.
Overall there appears to be a dose-dependent increase in efficacy & A/E, but otherwise oral minoxidil is well tolerated.
Adverse effect:
- Common – hypertrichosis
- Others – tachycardia, weight gain, hirsutism, lower extremity edema
- Rare – pericardial effusion, congestive heart failure, allergic reactions
4. Topicals
4.1 Topical Finasteride
- MOA: Local DHT inhibition (↓ conversion of testosterone → DHT) at the hair follicles.
- Advantage: fewer systemic effects compared to its oral counterparts
- Adverse effects: skin irritation & pruritus at the application site
4.2 Topical spironolactone
- Local inhibition of androgens on the hair follicle without the systemic side effect.
- Combination of minoxidil & spironolactone could be beneficial to patients.
c) Injectables
1. Platelet Rich Plasma (PRP) Treatment
MOA: The alpha granules within the platelets contain platelet-derived growth (PDGF), vascular endothelial growth factor (VEGF), transforming growth factor beta (TGFβ), and insulin-like growth factors (IGF) –> Induction of stem cell differentiation as well as promoting angiogenesis.
- PDGF : reduce follicular apoptosis & promoting follicular cell growth.
- IGF-1 : counteract the inhibitory effect of DHT on the hair follicle.
Treatment: PRP is injection into the deep dermis or subcutaneous tissue at a volume of 4 – 8 mL per session.
More effect in early-stage AGA.
Adverse effects (generally minimal), may include scalp sensitivity, mild scalp scaling, injection site pain & infection risk.
Contraindications:
- History of malignancy
- Platelet/bleeding disorders
- Anemia
- Pregnancy
- Immunocompromised
Limitations:
- Not curative → requires ongoing maintenance
- Lack of protocol standardization.
- Limited long-term evidence.
2. Exosomes/Stem cells
Emerging regenerative therapy using mesenchymal stem-cell derived exosomes. Acellular –> low immunogenicity risk. It is injected into the scalp & treatments may be spaced apart depending on the extent of hair loss. Side effects are minimal and may include minor pain at injection side, which usually resolves within 24 – 48 h.
d) Adjuvants
1. Microneedling
Minimally invasive. Used as an adjunct to standard treatments (e.g. minoxidil, finasteride).
Can be useful in treatment-refractory AGA.
MOA:
- Controlled repetitive, tissue microinjury –> transdermal microchannels within the stratum corneum –> promote growth factors, activate bulge stem cells, & increase expression of genes associated with hair growth.
- May also stimulate hair growth via upregulation of Wnt/B-catenin signaling & induction of VEGF.
- Can also enhance penetration of topical therapies.
Potential side effects may include :
- Pain during treatment
- Transient pinpoint bleeding
- Erythema
- Enlargement of lateral cervical lymph nodes
- Pruritis
- Seborrheic dermatitis
- Headache
- Risk of infection
2. Topical ketoconazole
Antifungal + anti-inflammatory (for Malassezia infection in seborrheic dermatitis)
Also has antiandrogenic properties with DHT inhibition.
Can be used OD/BD/ 2 – 3 x per week.
Shampoos containing 2% ketoconazole may be applied to the scalp as an adjuvant or alternative therapy in AGA treatment.
Limitation: No high-quality evidence for AGA efficacy. Otherwise, it has no significant side effects.
e) Supplements/Nutraceutical
1. Synergen Complex®
- Active ingredients include: saw palmetto, ashwagandha, cucumin, hydrolyzed marine collaged type I & III, palm extract (tocotrienol/tocopherol complex), horsetail, amino acids, black pepper fruit extract (piperine), Japanese knotweed, hyaluronic acid, and biotin.
- These ingredients inhibit 5-alpha-reductase, lower cortisol levels, reduce inflammation, promote homeostasis, and maintain collagen stores.
2. Marine complex supplement – e.g. Viviscal®, which contains proprietary blend of extracellular matrix components of shark & mollusks, vitamin C, horsetail extract, & flax seed extract.
3. Serenoa repens
- Active ingredient is saw palmetto, which is a palm tree berry extract that inhibits the 5-alpha reductase.
- Side effects are minimal, commonly gastric discomfort.
- Caution : it may reduce PSA levels by 50% after 6 – 12 months of treatment, thus possibly missing early detection of prostate cancer in patients self-medicating with Serenoa repens.
4. Plant-based oils – typically affordable & available OTC, however there are no FDA regulation or robust clinical studies.
- Rosemary oil: enhance micropapillary perfusion, increase prostaglandin E2 production, decrease leukotriene B4 production.
- Pumpkin seed oil: 5-AR antagonism
- Tea tree oil: anti-inflammatory & antimicrobial
Hair Transplantation
Last resort for patients who either failed medical therapy or have lost a significantly large & non-recoverable surface area of scalp hair that can only be treated by implanting new hairs into the area.
Combination therapy
Using combination therapy may provide better outcomes comparing to monotherapy by providing synergistic mechanisms to target the multiple disease pathways simultaneously. This can lead to greater improvement in hair density, count, and thickness, along with higher patient satisfaction & earlier visible results.
Common effective combinations may include :
- Topical Minoxidil + Finasteride – most common combination
- Topical Minoxidil + Microneedling
- LLLT + Minoxidil/Finasteride
- PRP + Minoxidil ± Microneedling (Triple therapy)
- For FPHL – Spironolactone + low-dose oral minoxidil
While combination therapy may provide better outcomes, it should be balanced against cost, complexity, & patient adherence.
Note
It is important to recognise that the management of androgenetic alopecia (AGA) is typically long-term. As such, factors including treatment cost, potential adverse effects, and patient preferences should be carefully considered.
A thorough discussion of the risks, benefits, and financial implications is essential to support informed, shared decision-making. Additionally, not all patients are significantly impacted by hair loss, and in such cases, treatment may not be necessary or desired.
References
- Nestor, M. S., Ablon, G., Gade, A., Han, H., & Fischer, D. L. (2021). Treatment options for androgenetic alopecia: Efficacy, side effects, compliance, financial considerations, and ethics. Journal of cosmetic dermatology, 20(12), 3759–3781. https://doi.org/10.1111/jocd.14537
- Ho CH, Sood T, Zito PM. Androgenetic Alopecia. [Updated 2024 Jan 7]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2026 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK430924/
- Kaiser, M., Abdin, R., Gaumond, S. I., Issa, N. T., & Jimenez, J. J. (2023). Treatment of Androgenetic Alopecia: Current Guidance and Unmet Needs. Clinical, cosmetic and investigational dermatology, 16, 1387–1406. https://doi.org/10.2147/CCID.S385861
- Redman, M. W., Tangen, C. M., Goodman, P. J., Lucia, M. S., Coltman, C. A., Jr, & Thompson, I. M. (2008). Finasteride does not increase the risk of high-grade prostate cancer: a bias-adjusted modeling approach. Cancer prevention research (Philadelphia, Pa.), 1(3), 174–181. https://doi.org/10.1158/1940-6207.CAPR-08-0092
